Application of safety switch in cell therapy
1、Safety switch of cell therapy
After autologous or allogeneic cell infusion, a large number of immune cells are activated and prolifed rapidly, resulting in excessive cascade release of cytokines such as IL-6, IL-1, IL-12, TNF-α, IFN-α, and GM-CSF, leading to symptoms such as fever, hypotension, capillary leakage, hypoxia, and end-organ dysfunction. Also known as cytokine release syndrome (CRS). In order to cope with the emergence of CRS in patients after receiving cell therapy, there are two approaches: on the one hand, predictive biomarkers of CRS and neurotoxicity can be detected, and timely response can be made through intensive monitoring; The other is to design safer cell therapies, and the consensus strategy is to put a "safety switch" on cells, treat them under human control, and step on the brake before adverse effects occur, which will help reduce the toxic side effects.
As technology has developed, it has become feasible to reversibly control cellular products by providing or removing small molecules, providing protein-based regulators, or by physical stimuli such as light, ultrasound, or heat [1].
2、Types of safety switches
(一)Leverage CID technology platform
CID stands for Chemical Induced Dimerization. The so-called CID technology platform (chemically induced dimerization technology) is to introduce specific DNA molecules into target cells so that the target cells express CID proteins (such as FKBP12) and target cells. The protein is then induced to dimerize by adding the small molecule compound CID, allowing the corresponding CID protein to exert its own function. CID protein includes a signal region and a binding region. Adding a dimerization inducer (such as rimiducid) can combine with the CID binding region to form a dimer, and the dimer will start the downstream signaling cascade reaction.
Caspase-9 is the initiating enzyme of the apoptosis signaling pathway. When activated, it will generate a signaling cascade and ultimately lead to apoptosis. In adoptive cell therapy, iCasp9 can be transduced to control the life activities of cells. iCasp9 is composed of the CID protein sequence through the linker and caspase9 sequences (Figure 1 B). When cells are exposed to dimerization-inducing drugs, it will cause cells expressing this structure to die rapidly, reducing severe side effects during treatment.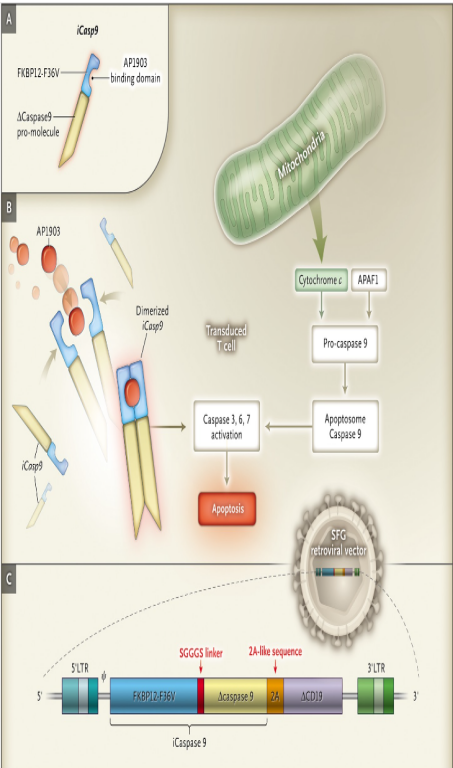
Figure 1. iCasp9 system regulates apoptosis [2]
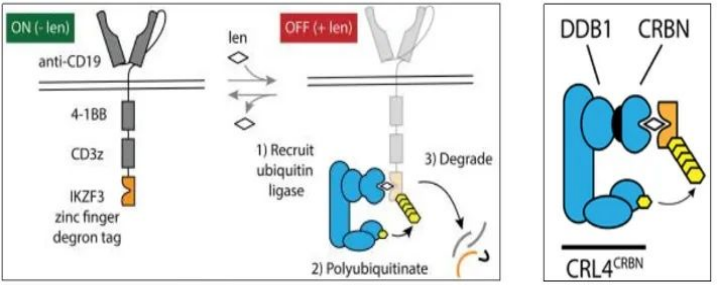
Figure 2. Lenalidomide regulates the degradation of CAR structure [3]
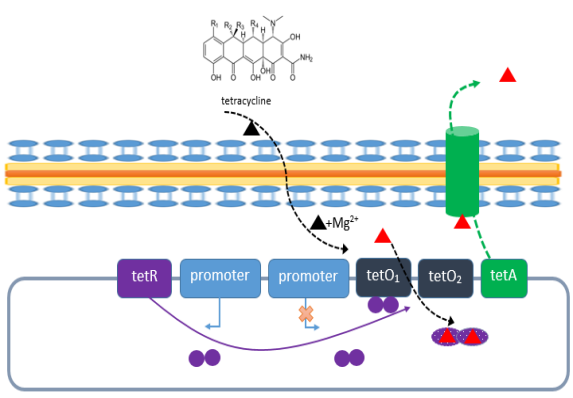
Figure 3. Tetracycline regulatory system of E. coli [4]
After autologous or allogeneic cell infusion, a large number of immune cells are activated and prolifed rapidly, resulting in excessive cascade release of cytokines such as IL-6, IL-1, IL-12, TNF-α, IFN-α, and GM-CSF, leading to symptoms such as fever, hypotension, capillary leakage, hypoxia, and end-organ dysfunction. Also known as cytokine release syndrome (CRS). In order to cope with the emergence of CRS in patients after receiving cell therapy, there are two approaches: on the one hand, predictive biomarkers of CRS and neurotoxicity can be detected, and timely response can be made through intensive monitoring; The other is to design safer cell therapies, and the consensus strategy is to put a "safety switch" on cells, treat them under human control, and step on the brake before adverse effects occur, which will help reduce the toxic side effects.
As technology has developed, it has become feasible to reversibly control cellular products by providing or removing small molecules, providing protein-based regulators, or by physical stimuli such as light, ultrasound, or heat [1].
2、Types of safety switches
(一)Leverage CID technology platform
CID stands for Chemical Induced Dimerization. The so-called CID technology platform (chemically induced dimerization technology) is to introduce specific DNA molecules into target cells so that the target cells express CID proteins (such as FKBP12) and target cells. The protein is then induced to dimerize by adding the small molecule compound CID, allowing the corresponding CID protein to exert its own function. CID protein includes a signal region and a binding region. Adding a dimerization inducer (such as rimiducid) can combine with the CID binding region to form a dimer, and the dimer will start the downstream signaling cascade reaction.
Caspase-9 is the initiating enzyme of the apoptosis signaling pathway. When activated, it will generate a signaling cascade and ultimately lead to apoptosis. In adoptive cell therapy, iCasp9 can be transduced to control the life activities of cells. iCasp9 is composed of the CID protein sequence through the linker and caspase9 sequences (Figure 1 B). When cells are exposed to dimerization-inducing drugs, it will cause cells expressing this structure to die rapidly, reducing severe side effects during treatment.

Figure 1. iCasp9 system regulates apoptosis [2]
(二)Using targeted protein degradation technologyA research team has created a molecular switch to regulate the activity of CAR-T cells, which can be achieved by administering a commonly used anti-cancer drug, lenalidomide [3]. They took advantage of a novel technology called targeted protein degradation, a pathway through which a handful of drugs, including lenalidomide, work by targeting the degradation of specific proteins. This mechanism was used to transform a small protein tag. When this degradation tag is attached to the CAR, during the administration of lenalidomide, lenalidomide will bind to the protein tag of the CAR structure and induce CRL4CRBN-mediated Through ubiquitination and proteasomal degradation, the tagged CAR will be degraded, thereby preventing T cells from recognizing cancer cells. Since T cells continue to produce CAR proteins, after lenalidomide is removed, the newly generated CAR proteins gradually accumulate, and CAR-T cells resume their anti-tumor functions.

Figure 2. Lenalidomide regulates the degradation of CAR structure [3]
(三) Utilizing tetracycline (Tet) regulatory systemSimply put, the tetracycline (Tet) regulated expression system controls the expression of the target protein by inducing drugs such as Tet to change the conformation of the regulated protein. In bacterial systems, under normal circumstances, the tetracycline repressor protein (tetR) will bind to the tetracycline resistance operon (tetO) to inhibit the transcription of the downstream resistance gene tetA. When tetracycline or tetracycline analogs such as doxycycline (Dox) are present, tetR will bind to tetracycline and no longer bind to tetO, causing the expression of the downstream resistance gene tetA. The tetracycline will be transported out of the cell through the tetA protein, and the bacteria will Acquire drug resistance [4].

Figure 3. Tetracycline regulatory system of E. coli [4]
According to characteristics, it can be divided into: activating system Tet-on and inhibitory system Tet-off.
Tet-off principle: tTA is a protein fused from TetR and viral transcriptional activation domain VP16. When Dox is not present, tTA binds to TRE (seven repeated TetO sequences) to initiate downstream gene expression; when Dox is present, the conformation of tTA changes, and tTA will fall off the TRE, causing downstream gene expression to shut down.
Tet-on principle: rtTA is a protein fused by rTetR and VP16. Its phenotype is opposite to that of tTA. rtTA is an anti-Tet repressor (rTetR) and relies on the presence of tetracycline for induction, rather than repression. When Dox is absent, rtTA cannot bind to TRE, and downstream gene expression is shut down. When Dox is present, the conformation of rtTA changes, rtTA gathers TRE, and downstream gene expression is turned on.
The tetracycline regulatory system can be used to precisely regulate the expression of target genes to control whether cells express CAR structures or to regulate cell death.
Reference
[1]Sahillioglu, A.C. and T.N. Schumacher, Safety switches for adoptive cell therapy. Curr Opin Immunol, 2022. 74: p. 190-198.
[2]Di Stasi, A., et al., Inducible apoptosis as a safety switch for adoptive cell therapy. N Engl J Med, 2011. 365(18): p. 1673-83.
[3]Jan, M., et al., Reversible ON- and OFF-switch chimeric antigen receptors controlled by lenalidomide. Sci Transl Med, 2021. 13(575).
[4]https://2013.igem.org/Team:Bielefeld-Germany/Biosafety/Biosafety System M.
Disclaimer: Shenzhen Cell Valley is committed to the research of cell and gene therapy, in order to promote emerging technologies and let more people understand the new developments in biomedicine.The content of this article is for information exchange only. This platform remains neutral with respect to the content, statements, and opinion judgments in the article, and does not represent the position and opinions of Shenzhen Cell Valley.The relevant information in this article should not be used for diagnosis or treatment, and cannot replace professional medical advice. Our website will not assume any responsibility.The final interpretation of the above statement belongs to our company’s website. This statement will apply to articles shared on our website at all times. Thank you for your cooperation! Copyright statement: The copyright of the article belongs to Shenzhen Cell Valley. Individuals are welcome to forward it to friends, media or Any unauthorized reproduction by the organization to other platforms will be regarded as infringement.If you need to reprint, please contact email: contact@sz-cell.com
Tet-off principle: tTA is a protein fused from TetR and viral transcriptional activation domain VP16. When Dox is not present, tTA binds to TRE (seven repeated TetO sequences) to initiate downstream gene expression; when Dox is present, the conformation of tTA changes, and tTA will fall off the TRE, causing downstream gene expression to shut down.
Tet-on principle: rtTA is a protein fused by rTetR and VP16. Its phenotype is opposite to that of tTA. rtTA is an anti-Tet repressor (rTetR) and relies on the presence of tetracycline for induction, rather than repression. When Dox is absent, rtTA cannot bind to TRE, and downstream gene expression is shut down. When Dox is present, the conformation of rtTA changes, rtTA gathers TRE, and downstream gene expression is turned on.
The tetracycline regulatory system can be used to precisely regulate the expression of target genes to control whether cells express CAR structures or to regulate cell death.
Reference
[1]Sahillioglu, A.C. and T.N. Schumacher, Safety switches for adoptive cell therapy. Curr Opin Immunol, 2022. 74: p. 190-198.
[2]Di Stasi, A., et al., Inducible apoptosis as a safety switch for adoptive cell therapy. N Engl J Med, 2011. 365(18): p. 1673-83.
[3]Jan, M., et al., Reversible ON- and OFF-switch chimeric antigen receptors controlled by lenalidomide. Sci Transl Med, 2021. 13(575).
[4]https://2013.igem.org/Team:Bielefeld-Germany/Biosafety/Biosafety System M.
Disclaimer: Shenzhen Cell Valley is committed to the research of cell and gene therapy, in order to promote emerging technologies and let more people understand the new developments in biomedicine.The content of this article is for information exchange only. This platform remains neutral with respect to the content, statements, and opinion judgments in the article, and does not represent the position and opinions of Shenzhen Cell Valley.The relevant information in this article should not be used for diagnosis or treatment, and cannot replace professional medical advice. Our website will not assume any responsibility.The final interpretation of the above statement belongs to our company’s website. This statement will apply to articles shared on our website at all times. Thank you for your cooperation! Copyright statement: The copyright of the article belongs to Shenzhen Cell Valley. Individuals are welcome to forward it to friends, media or Any unauthorized reproduction by the organization to other platforms will be regarded as infringement.If you need to reprint, please contact email: contact@sz-cell.com


